FSHD 101
Facioscapulohumeral muscular dystrophy or FSHD is a highly complex, progressive muscle wasting disease. It does not discriminate, affecting the lives of men, women and children from all walks of life. The Global footprint of this condition is enormous. Worldwide, FSHD affects the lives of an estimated 1 million people, a figure that does not take into account the effect FSHD has on the friends and family of the person affected.
Despite being considered one of the most common forms of muscular dystrophy in adults and children there are no treatments and no cure.
FSHD is commonly associated with progressive weakening of facial, shoulder and upper arm muscles. However, this explanation does little justice to a disease that can rob people of their ability to walk, talk, smile or even eat. The regression often comes in bursts with sudden deterioration followed by periods of no change.

The loss of skeletal muscle has a huge impact on daily life making even simple tasks complicated. Living with FSHD means living with pain, fatigue and the social isolation that comes from being reliant on mobility aids. The future for someone with FSHD is uncertain because there is so much variability in how FSHD manifests in people. People with FSHD live with no known cure and few treatments currently available. The FSHD Global Research Foundation is working to change this and gives hope to those living with this disease that something is being done to fight for a cure.
GENETIC MECHANISM OF FSHD
FSHD is arguably the most complex genetic condition currently known. FSHD involves a convoluted and complicated interplay between genes and proteins in the muscle cell.
While a massive amount of progress has occurred resulting in an improved understanding of the genetic mechanism for FSHD, we are still trying to completely understand how the genetic mutation causes the range of symptoms experienced by people with FSHD. Below is outlined the mechanism for FSHD1 and FSHD2.
At the moment there are researchers investigating an FSHD type 3 and it is likely that more subtypes of FSHD will be identified as our knowledge of FSHD improves.
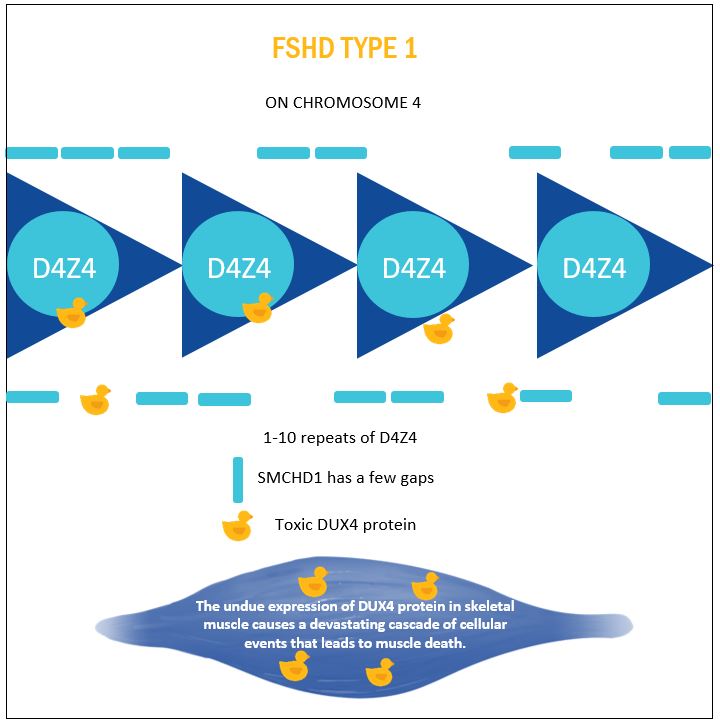
FSHD TYPE 1
FSHD is unlike most genetic conditions where a mutation causes pathological changes in a particular gene and protein. FSHD is caused by mutations that actually increase the expression of a toxic protein.
Nearly all cases of FSHD are associated with a mutation on chromosome 4. Chromosome 4 contains a series of repeated pieces of DNA, so called D4Z4 units. People without FSHD1 have 11 – 100 D4Z4 units. In people with FSHD 1 (95% of cases) the D4Z4 array is shortened to 1 – 10 units.
The D4Z4 units act like a lock for this region of the genome. With fewer repeats a gene embedded in this region called DUX4 is expressed. The unwanted production of the DUX4 protein, sets in motion a cascade of events that leads to loss of muscle function. The exact mechanisms are still being studied, however it appears that DUX4 expression causes muscle cell damage. This loss of cells causes muscles to waste away. DUX4 may also damage the normal muscle regeneration pathways preventing muscles from repairing any damage experienced through normal daily activities. DUX4 is also thought to invoke an immune response further inflicting damage on the muscles. Oxidative stress is also thought to play a role. As we learn more about the role of DUX4 the exact mechanisms for FSHD will become clearer.

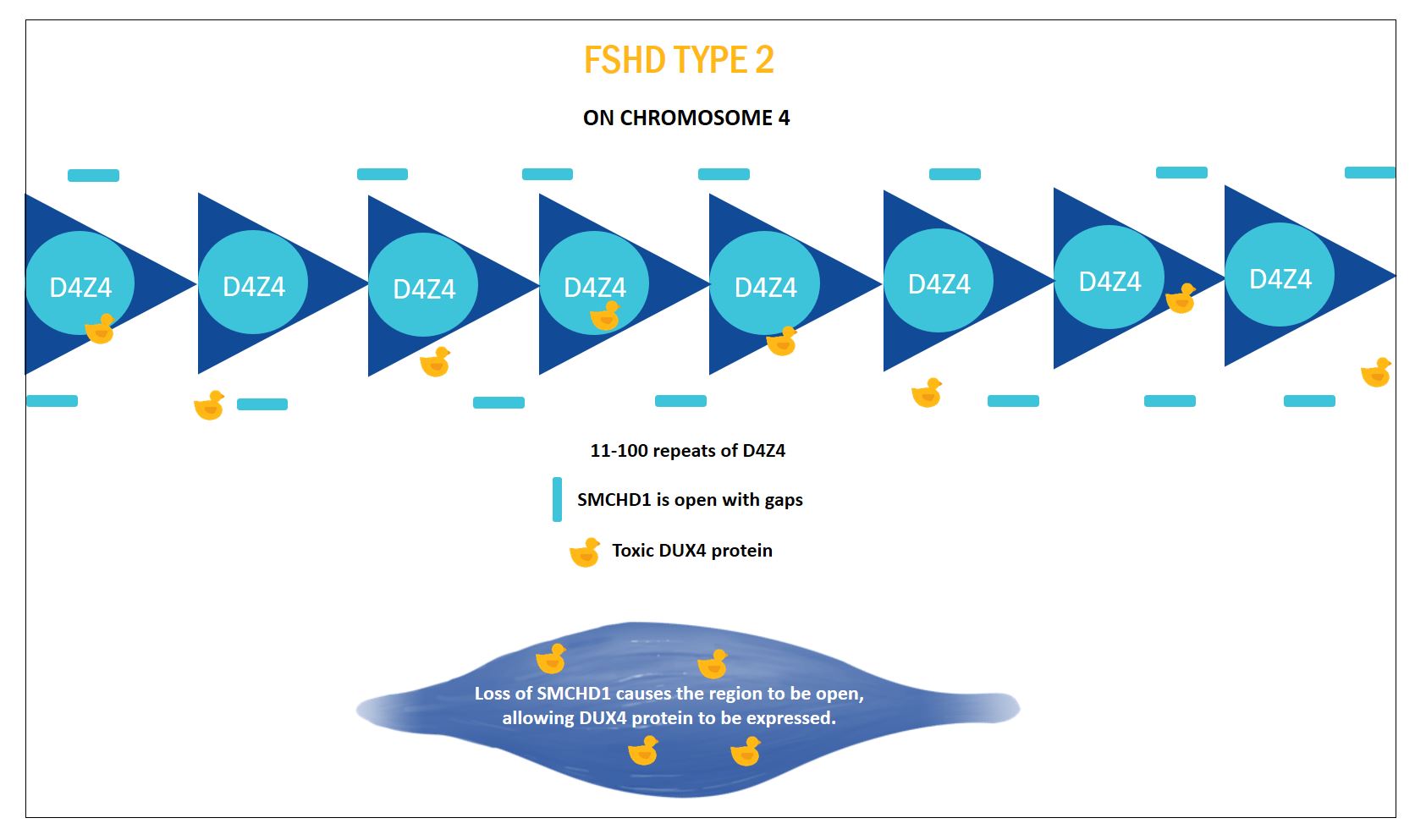
FSHD TYPE 2
FSHD type 2 was characterised when a large family was found with FSHD symptoms indistinguishable from FSHD1, but without the contraction of the D4Z4 domain on chromosome 4. People with FSHD2 (5% of cases) have more than 11 D4Z4 units like people without FSHD.
The defect in FSHD2 was found to be in a gene called Structural Maintenance of Chromosomes Hinge Domain Containing 1 (SMCHD1). This gene acts as a lock for regions on the genome, in people without FSHD this protein is keeping regions like the one that contains the DUX4 protein, closed. Mutations in SMCHD1 lead to a smaller amount of this protein being produced. Less protein means less repression on regions of the genome that are usually closed. This includes the D4Z4 region causing DUX4 to be expressed where it is able to cause the same types of damage to muscle cells observed in FSHD1.
FSHD Global will continue to update this page as discoveries are made. Research in this area is progressing very quickly. If you would like to be kept up to date on new research in FSHD please subscribe to our Monthly newsletter ‘Progress’. This newsletter contains summaries of recently published research and updates from FSHD Global funded research along with updates from the FSHD Global community.